Abstract
DLBCL is the most common type of non-Hodgkin lymphoma in adults. Although frontline standard of care treatment is effective in 60-70% of patients, 10-15% have primary resistance and 20-30% will relapse within 18 months. There is an unmet need to predict treatment refractoriness and relapse in order to intensify treatment approaches early during induction therapy. Early molecular response (EMR), defined as the dynamics of ctDNA after one cycle of immunochemotherapy, was shown to be prognostic in a small cohort of first-line DLBCL patients (Kurtz et. al. J. Clin. Oncol 2018), and recently validated using samples from the POLARIX study (NCT03274492) in previously untreated DLBCL.
To support clinical studies, the Roche Molecular CAP/CLIA Laboratory has developed and validated the AVENIO Oncology Assay Non-Hodgkin Lymphoma Test (AOA NHL Test) that uses next generation sequencing of plasma cell-free (cf)DNA and of matched genomic (g)DNA to identify tumor-specific single nucleotide variants (reporter SNVs) in a panel of > 400 genomic regions and to monitor the dynamics of the reporter SNVs on treatment using a Monte-Carlo p-value statistical algorithm (Newman et. al. Nat Biotechnol 2016). The AOA NHL Test determines ctDNA presence or absence as an indicator of MRD, and assigns high-risk or low-risk EMR status in patients with DLBCL by measuring changes in ctDNA burden on treatment and applying a previously established cutoff.
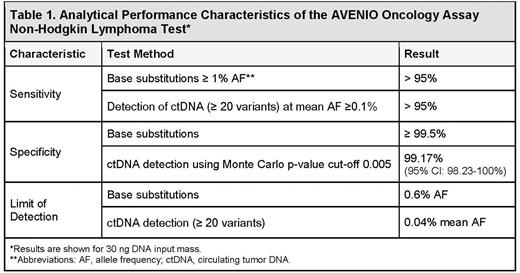
The analytical validation studies for the AOA NHL Test were designed to address accuracy, sensitivity, specificity, linearity, and precision of the following 1) variant detection in plasma samples; 2) ctDNA detection (measured as mean allele fraction (AF) of reporter SNVs using a Monte-Carlo p-value statistical algorithm); and 3) classification accuracy of the log-fold change (LFC) of mean mutant molecules per mL of plasma (MMPM), using clinically relevant cutoffs. Validation samples consisted of Genome in a Bottle blended gDNAs, serial dilution of healthy donor cfDNA blends, and serial dilution of DLBCL ctDNA into healthy donor cfDNA. Key results of the analytical validation are shown in Table 1.
In summary, analytical validation of the AOA NHL Test demonstrated high sensitivity for detection of individual variants and ctDNA, and the capability of accurately detecting changes in ctDNA levels as measured by LFC in MMPM. The assay sensitivity improves with the increase in cfDNA input mass as well as the number of reporter variants while specificity (i.e. ability to detect signal over noise) can be maintained from sample to sample by pre-selecting a Monte-Carlo p-value cutoff. The final recommended sample acceptance criteria as well as sequencing QC metrics will be presented.
Of 858 cases of DLBCL patients examined using the initial assay prototype and the validated AOA NHL Test, including samples from the POLARIX biomarker validation study, 846 (98.6%) had identifiable reporter variants in pre-treatment plasma samples with a median number of reporter SNVs of 137 per patient (range 1-1039), demonstrating that almost 100% of patients can be monitored with this assay. Of the 12 patients with no detectable reporters, 11 had less than 10 ng cfDNA available, highlighting the importance of sufficient sample input criteria established by analytical validation. The test is currently being used in a global prospective Phase 2 clinical trial (NCT04980222) to test the safety and efficacy of glofitamab in combination with R-CHOP in patients with High Risk EMR status.
Acknowledgements: Alex Lovejoy for advice on the analytical validation design, Suzanne Cheng, Jennifer Holmstrom and Jill Ray for program management, and Bowdoin Su and Yanwen Jiang for abstract preparation.
Disclosures
Stokowski:Roche Molecular Diagnostics: Current Employment, Current equity holder in publicly-traded company. Tabari:Roche Molecular Diagnostics: Ended employment in the past 24 months. Bogard:Roche Molecular Diagnostics: Current Employment. Hacker:Roche Molecular Diagnostics: Current Employment, Current equity holder in publicly-traded company. Kameneva:Roche Molecular Diagnostics: Ended employment in the past 24 months. Ji:Roche Molecular Diagnostics: Ended employment in the past 24 months. Teng:Roche Molecular Diagnostics: Current Employment. Melnikova:Roche Molecular Diagnostics: Current Employment, Current equity holder in publicly-traded company. McCord:Genentech: Current Employment, Current equity holder in publicly-traded company. Punnoose:Genentech: Current Employment, Current equity holder in publicly-traded company. Loberg:Roche Molecular Diagnostics: Current Employment, Current equity holder in publicly-traded company. Shabbeer:Roche Molecular Diagnostics: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.